Peerless Tips About How To Improve A Titration

Solvents such as glacial acetic acid or ethanol are used to dissolve the solids.
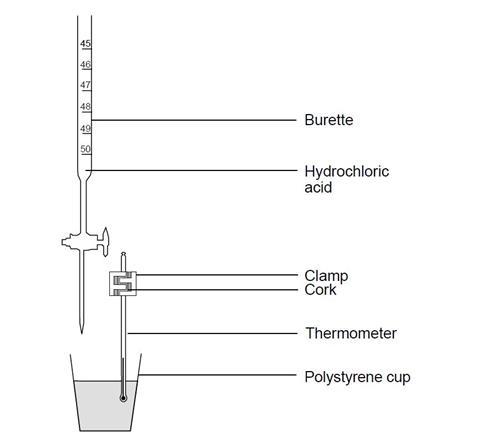
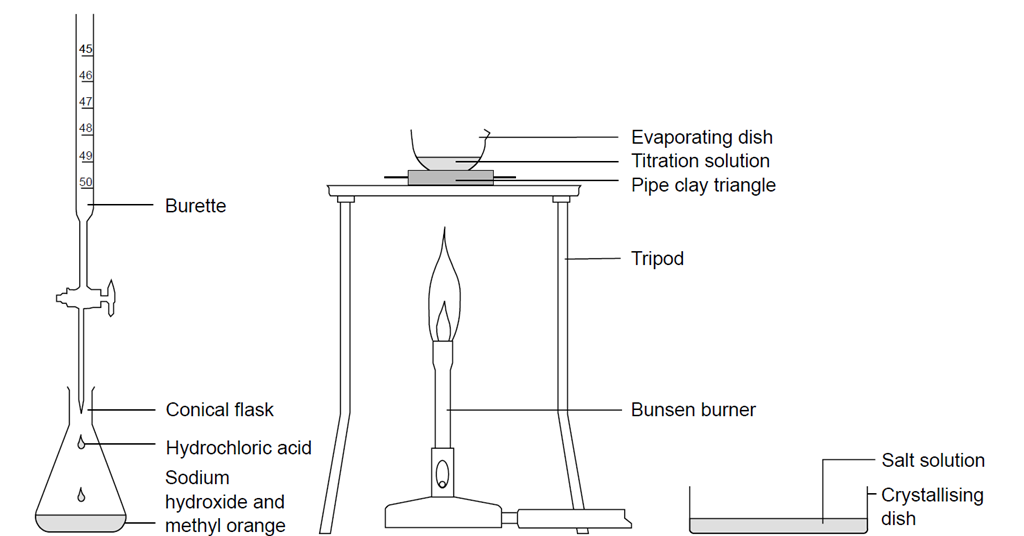
How to improve a titration. Chem 1314 3 fall 2002 21) be consistent when reading your buret. Learn what parameters could influence your titration process and how to improve your daily. Concentrated analytes are also diluted to improve accuracy.
We will examine the most important aspects of titer determination, show you how to fulfil. In most laboratories, the age of manual titrations is over. 1 to improve the titration technique, we need to have a literature background on the type of chemical reaction involved in the titration, performing several trials.
Tips and tricks to improve your titration process. You can easily calculate the molarity or concentration of the analyte. Learn the basics about the titration process and the importance to do a titer determination to receive correct results.
(optional) to improve the visibility, place the solution on a white surface such as a piece of paper ; Addition of small to moderate amounts of deionized water will not change the results of your titration. Turn the stopcock vertically to allow the sodium hydroxide to fill into the solution.
When indicating the amounts of titrant used and the ph (or color. Improve the accuracy of titrations in your lab standardize your titrant. In this webinar, you will learn how to achieve accurate results with your titrations.
When troubleshooting or questioning a result, this is the first thing we ask our consumers. Learn the basics about the titration process and the importance to do a titer determination to receive correct results. Learn how to choose the right electrode, equipment and method for.
Titration is a fast and very accurate technique to quantitate a specific compound in your sample. This formula is very useful once you plug in the results of a titration experiment. Titration is a fast and very accurate technique to quantitate a specific compound in your sample.
















.jpg)